Description
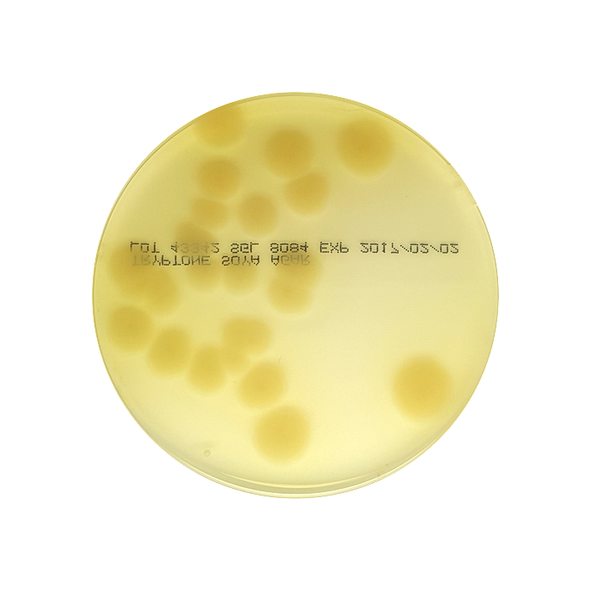
This is a general-purpose complex medium for cultivation and isolation of fastidious bacteria, yeasts and moulds.
The formulation is based on the United States Pharmacopoeia (USP Medium II) and European Pharmacopoeia (EP Medium B). The medium can be incubated under aerobic or anaerobic conditions for sterility testing, air sampling and other areas of bacteriological investigation.
This product is wrapped in barrier film to allow for use in Vaporised Hydrogen peroxide sterilisation systems.
This particular product is triple wrapped and terminally sterilised by Gamma irradiation. Dose Range: 8.0 kGy ? 15.0kGy